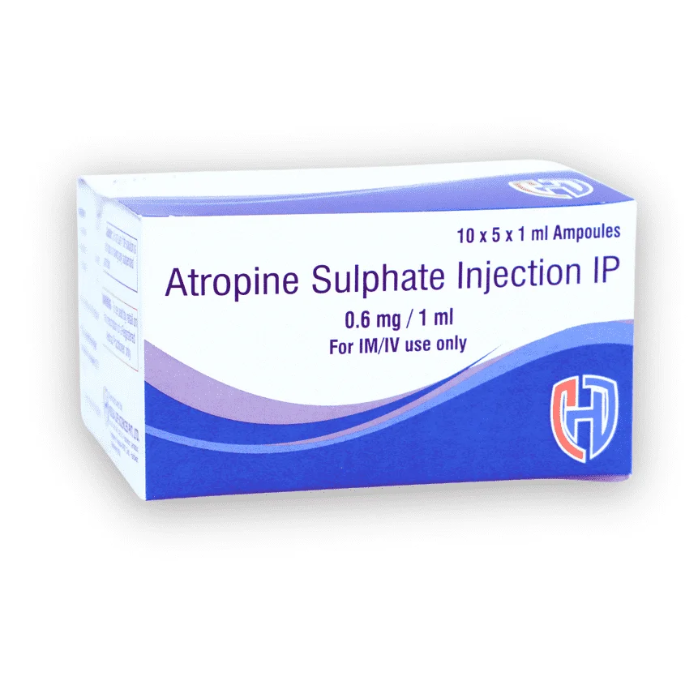
Atropine Sulphate Injection IP 0.6mg/1ml is a critical care injectable used for a variety of emergency and surgical applications, including bradycardia management and as a pre-anesthetic agent. Manufactured by Heilsa Life, a reputed pharmaceutical company based in Roorkee, Uttarakhand, India, this product is widely trusted for its clinical efficacy and compliance with global quality standards.
Formulated and packed in 10 x 5 x 1ml ampoules, this injectable is suitable for intramuscular (IM) and intravenous (IV) use. We are open to third party manufacturing, contract production, and global exports, catering to pharma marketing companies, hospitals, and distributors worldwide.
Our facility adheres to WHO-GMP, ISO 9001:2015, and stringent regulatory norms, making us a reliable partner for clients seeking custom packaging, documentation support, and consistent supply.
Why Partner with Heilsa Life?
-
Located in Roorkee, Uttarakhand, a trusted pharma hub in India
-
WHO-GMP certified production facility
-
Experienced in export to over 25 countries
-
Flexible third party manufacturing and bulk export options
-
Transparent documentation and regulatory assistance
-
Reliable, scalable production with strict quality control
Frequently Asked Questions (FAQs):
Q1. What is Atropine Sulphate Injection used for?
It is commonly used to treat bradycardia, reduce salivation before surgery, and as an antidote for chemical poisoning.Q2. Can you offer this under our brand name?
Yes, we offer third party/private label manufacturing as per your brand specifications.Q3. Are your products certified?
Yes, we are WHO-GMP and ISO certified, with all necessary documentation available for export.Q4. What is the shelf life of this injection?
Generally, 24 to 36 months depending on storage and packaging. Custom shelf life labeling available.Q5. Do you export internationally?
Yes, we export to Asia, Africa, Latin America, and more with full support for registration and logistics.
Contact Us:
📍 Heilsa Life Sciences
- 🏠7 KM Milestones, Roorkee – Chandigarh Highway, Puhana Chowk, Roorkee – 247667, Haridwar, Uttarakhand
📞 Phone: [+91-9258237506]
📧 Email: info@heilsalife.com
🌐 Website: www.heilsalife.com


Reviews
There are no reviews yet.